Remember this commercial for Neuriva, a recently introduced brain supplement?
The claims in it that Neuriva has “clinically proven ingredients that fuel five indicators of brain performance” became part of a class action lawsuit against the company. The complaint asserted that Reckitt Benckiser, the maker of Neuriva, lacked scientific proof that the product really improves brain functioning.
A review of the relevant scientific literature shows that no valid scientific or clinical evidence exists regarding how much, if any, of Neuriva’s key ingredients reaches the brain. Because of this lack of evidence, Defendants’ claims that Neuriva’s ingredients are scientifically and clinically proven to benefit the brain or enhance brain performance are patently false, as well as are Defendants’ claims that Neuriva has been scientifically proven to be effective. Indeed, no publicly available study of Neuriva exists, and Plaintiffs have found no indication that Neuriva’s efficacy has ever been studied or tested.
The company denied the charges but nonetheless subsequently entered into a proposed settlement agreement which calls for full or partial refunds to consumers and a change in the claims the company makes on the packaging and in advertising. [Purchasers can file a claim here, although the settlement is not finalized yet.]
*MOUSE PRINT:
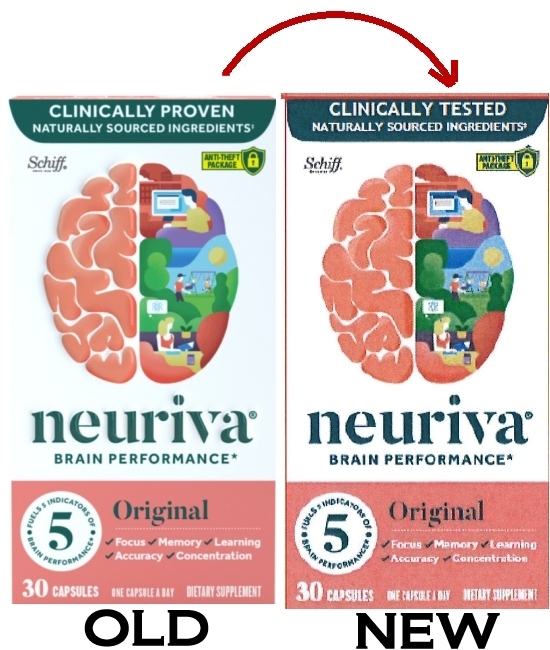
Put simply, the company agreed to change the word “proven” to “tested” along with other minor wording changes, but only for a period of two years. Frankly, whether it says “clinically proven” or “clinically tested” I think most consumers will still come away with the same net impression that there is reliable scientific evidence backing up the brain performance claims. The proposed settlement has also come under fire from other consumer advocates and court watchers.
The product’s TV advertising has changed already. They now have Miyam Bialik, the actress best known for her roles on Blossom and The Big Bang Theory, and as the future host of Jeopardy!, vouching for the product. What qualifies her for this role? She got her doctorate in neuroscience. Now the company uses that fact to also claim that the product is “neuroscientist approved.”
One thing that hasn’t changed is a disclaimer on the side of the Neuriva package.
*MOUSE PRINT:




